Hi all! Rabia here, I came across an intriguing paper highly relevant to my work on the rapid in vivo validation of HDAC inhibitor-based treatments using neuroblastoma zebrafish xenografts. The study outlines a zebrafish neuroblastoma yolk sac model specifically designed to evaluate both the effectiveness and toxicity of histone deacetylase (HDAC) inhibitors.
HDAC inhibitors are drugs that target specific enzymes involved in gene regulation. This study tested broad-spectrum HDAC inhibitors as standalone treatments and combined them with doxorubicin, a well-known chemotherapy drug.
But why on Zebrafish? The zebrafish model provides a rapid and efficient means of testing these treatments, offering valuable insights into their potential use in combating neuroblastoma. This model allows for assessing drug efficacy and helps understand the associated toxicities quickly, making it a powerful tool for developing new anti-cancer therapies.
In the study, fish larvae were implanted with fluorescently labelled, well-established neuroblastoma cell line (SK-N-BE(2)-C) and patient samples (HD-N33, NB-S-124) to grow tumours. Non-cancerous cells (VH7 fibroblasts) were utilized to verify that tumour progression in zebrafish was specific to tumour cells. The engraftment of human cells into fish larvae was confirmed by immunohistochemistry (IHC) staining on zebrafish sections injected with neuroblastoma cells (SK-N-BE). This was achieved using a STEM121 antibody that reacts specifically with a human cytoplasmic protein. The findings showed that pediatric tumour cells survive and grow in the zebrafish model at rates like those observed in human tumours.
Before testing drug efficacy in zebrafish xenografts, optimal drug concentrations and maximal tolerated doses (MTD) were determined. Toxicity tests were conducted by treating fish larvae cells for three days without tumour cell injection to identify the maximum tolerated dose that did not cause observable morbidity, changes in morphology, or severe aberrations in larval behaviour. and lethal dose (LD) for each compound. To find optimal drug concentrations, larvae with xenografted tumour cells were incubated with increasing drug doses 24 hours post-implantation to the maximally tolerated dose (MTD). The relative IC50 values were then determined based on changes in tumour mass volume.
To evaluate the treatment, SK-N-BE(2)- cells were used to test the broad-spectrum HDAC inhibitors, including panobinostat, vorinostat, and tubastatin A, both alone and combined with doxorubicin. The partial response rate (PR) was measured to see how well different drug combinations work to shrink tumours in the zebrafish model. Here’s what they found: Doxorubicin combined with panobinostat resulted in a 23% PR, Doxorubicin combined with tubastatin A showed a 31% PR, and Doxorubicin combined with vorinostat achieved the best result with a 36% PR.
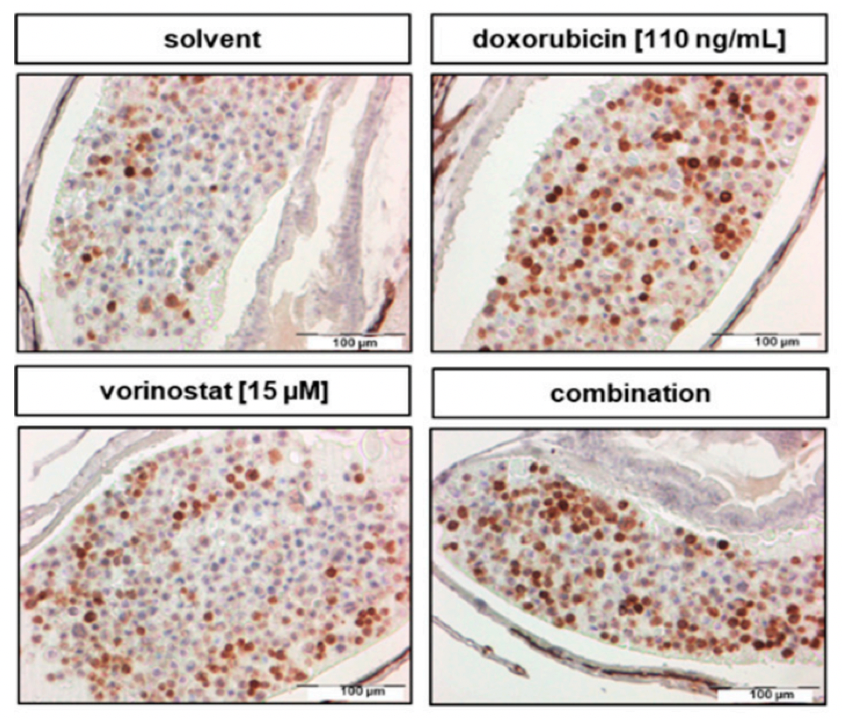
To test the effectiveness of the HDAC inhibitor treatment, they monitored the tumour growth using a confocal microscope before and 48 hours after giving the drug. The test revealed that a 48-hour treatment of SK-N-BE (2)-C zebrafish xenografts with vorinostat and doxorubicin alone, `and in combination, increased cell death. The combination of these two drugs was the most effective, causing a significant increase in cancer cell death (apoptosis) by decreasing cell proliferation, as indicated by reduced PPH3 marker and activating the number of Cleaved caspase-3 (Figure 1).
In essence, this study validates the use of HDAC inhibitors in treating neuroblastoma and paves the way for broader applications of zebrafish models in cancer research. As we look to the future, these innovative models could significantly enhance our ability to develop effective cancer therapies, making strides towards better treatments and, ultimately, more effective cures.
Written by Rabia Saleem