Finding suitable research models to study disease is a big challenge for researchers around the world. In cancer research, it is essential to work with models that can recapitulate tumour characteristics as much as possible. This is important to test chemotherapeutic drugs, understand tumour behaviour and have higher chances of translating the finds from the laboratory to clinical practice.
Multiple factors influence tumour behaviour and disease progression. The most important is the tumour microenvironment, which comprises different cells and molecules that surround the tumour and the extracellular matrix, a network of molecules that provides support to the cells in the body.
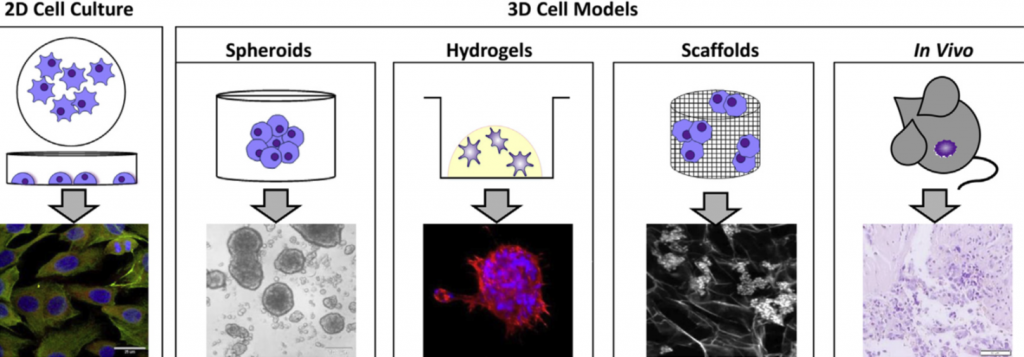
Most cell studies in a laboratory are based on 2D cell culture models in which the cells grow in a monolayer. Although this approach has a low cost and it is easy to use, it lacks the complexity observed in the clinical scenario. It is true that no model can recapitulate all the complexity found in the body. However, scientists were able to develop interesting approaches to study different tumour characteristics with relatively good approximation1.
Specifically for neuroblastoma, the most common solid tumour that affects children, scientists developed 3D models in which neuroblastoma cells grow interacting with the surrounding environment and with each other in a vial. Examples of 3D models include cells grown in hydrogels or scaffolds and multicellular tumour spheroids (see image below). Spheroids are formed through the self-adhesion of tumour cells growing in the form of very small balls. They can be maintained in the laboratory on their own or supported by scaffold-based platforms (jelly-like or porous materials). Scaffolds essentially support the cell resembling the extracellular matrix and surrounding tissue in the body.
In the Cancer Bioengineering Research Group, we work with neuroblastoma models such as organoids, a more complex type of spheroid, to understand neuroblastoma migration and invasion2. Moreover, we recently shared with the research community a protocol at jove.com describing the development of a 3D neuroblastoma model using collagen-based scaffolds3.
These models have the potential to advance drug tests performed in the laboratory providing better clinical translation, ultimately contributing to improving the quality of life and survival of children diagnosed with neuroblastoma.
The work with 3D models at the Cancer Bioengineering Research Group is supported by the Irish Research Council, the Conor Foley Neuroblastoma Cancer Research Foundation, Neuroblastoma UK and National Children’s Research Centre.
Written by Luiza Erthal
References
1. Nolan, J. C. et al. Preclinical models for neuroblastoma: Advances and challenges. Cancer Lett. 474, 53–62 (2020).
2. Gavin, C. et al. Neuroblastoma Invasion Strategies Are Regulated by the Extracellular Matrix. Cancers 13, 736 (2021).
3. Gallagher, C., Murphy, C., O’Brien, F. J. & Piskareva, O. Three-dimensional In Vitro Biomimetic Model of Neuroblastoma using Collagen-based Scaffolds. J. Vis. Exp. 62627 (2021) doi:10.3791/62627.